 For example, the orbital angular quantum number \(l\) can never be greater or equal to the principal quantum number \(n(l < n)\). The relative abundance of Deutrium (1 proton, 1 neutron) is so small that it is barely accounted for when calculating the average atomic mass. Protium is by far the must abundant isotope of hydrogen, and it only contains 1 proton, and no neutrons. Not all sets of quantum numbers (\(n\), \(l\), \(m\)) are possible. The average atomic mass for hydrogen is actually around 1.008 amu. 
Therefore, Relative atomic mass average mass of an atom/mass of carbon-12 x (1/12) 1.0076632 u / 1 u 1. As we saw earlier, it is convenient to use a reference unit when dealing with such small numbers: the atomic mass unit. Average mass of hydrogen (1.007825 u x 99.98) (2.014101 u x0.02 ) (1.007623 0.0000402) u 1.0076632 u The value of one twelfth of the mass of carbon-12 isotope is 1 u. a pollution of 1 gram of benzene in a certain amount of water converts to N A/78.11≈ 7.(\theta)\Phi_m(\phi). We now know that a hydrogen atom has a mass of 1.6735 x 10-24 grams, and that the oxygen atom has a mass of 2.6561 X 10-23 grams. Using the above calculator you could find that e.g. Or 1 mole of a substance will contain Avogadro's number of that substance. A hydrogen-like atom consists of one nucleus of charge Ze and a single electron of charge e, so in addition to H we have He . The term " mole" is defined in that one mole of a substance with a molecular (or atomic) mass of one (1), will have a mass of 1 gram. It is defined to be 1/12 of the mass of one atom of carbon-12 and in older works is also abbreviated as "amu".Īlso, important in this field is Avogadro's number (N A) or Avogadro's constant (6.0221 x 10 23). In related terms, another unit of mass often used is Dalton (Da) or unified atomic mass unit (u) when describing atomic masses and molecular masses. Molecular mass or molar mass are used in stoichiometry calculations in chemistry. This Calculator has been tested on Internet Explorer version 6 only,įirefox might not show all fields correctly. 
For question or remarks please contact us. !!! Lenntech BV cannot be held responsible for errors in the calculation, Make sure you enter the molecule of crystallization at last (e.g. The calculator handles at most two different bracket levels. The molecular mass calculator will recognize the entered formula's, which are included in the list of organic compounds. We will treat the proton and electron as point masses that interact. Or you can choose by one of the next two option-lists, which contains a series of common organic compounds (including their chemical formula) and all the elements. The hydrogen atom is a two-particle system consisting of a proton and an electron. An atom which has 9 protons, 8 neutrons, and 9 electrons will have an atomic mass of. 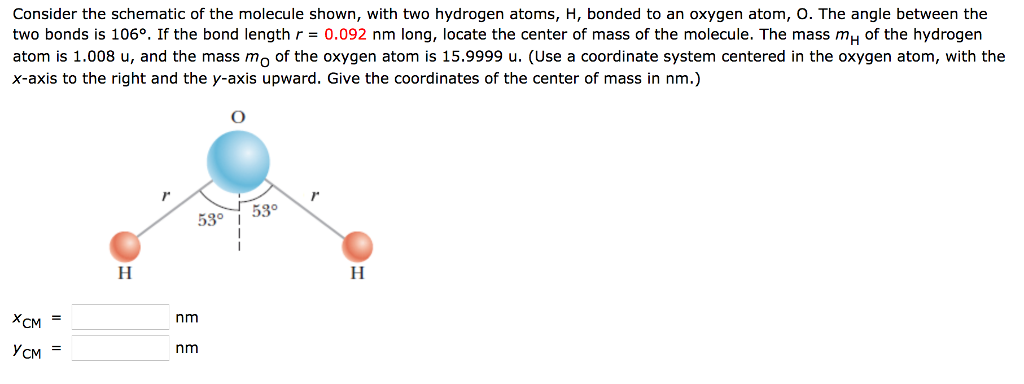
Add up the atomic masses of the Steps for. Other diatomic molecules include chlorine and hydrogen.11. 
This online calculator you can use for computing the average molecular weight (MW) of molecules by entering the chemical formulas (for example C3H4OH(COOH)3 ). (This formula only means the hydrogen peroxide molecule contains equal numbers of hydrogen and oxygen atoms. Molecular Weight Calculator Molecular Weight Calculator To find how much one atom weighs, we must use.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
